
Why sensor manufacturers are choosing MIPs for their next devices
ARTICLE
Sensors play a huge, yet often un-noticed role in modern society. If you drive a car, chat on the phone or cook with your oven, then you are interacting with sensors on a daily basis. These types of sensors detect movement, noise, or temperature, but there are also more complex sensors that detect biomolecules; biosensors. Biosensors are devices used to detect the presence or concentration of a biological analyte (typically in very small concentrations), such as a biomolecule or a microorganism. Biosensors consist of three parts: the receptor, that recognizes the biological analyte and produces a signal, a signal transducer, and a reader device. The receptor is traditionally a biological element such as an enzyme, antibody or nucleic acid. Molecularly Imprinted Polymers (MIPs) are alternative receptors that are completely synthetic, and offer a range of properties that natural receptors cannot match. Read on to learn what MIPs are and why their unique properties are making them the new receptor of choice for sensor manufacturers.
First things first, what are Molecularly Imprinted Polymers (MIPs)?
Molecularly imprinted polymers, also termed synthetic antibodies, are polymers that have been produced in a way to selectively bind to a molecule of interest. The molecule of interest can range from a small molecule such as caffeine, through to peptides, proteins and even whole viruses, such as COVID-19. They can be produced using several methodologies (1), however solid phase synthesis is proving to be one of the more popular methods to obtain MIPs with a high affinity and specificity to their target. This method produces MIP nanoparticles with a single binding site that act as antibody mimics (nanoMIPs). In this method, visualized in Figure 1, the target molecule is immobilized in a specific orientation on a solid phase support, and controlled polymerization is carried out around the target molecule using pre-selected monomers. A series of elution steps are then carried out to detach and isolate the high affinity nanoMIPs.
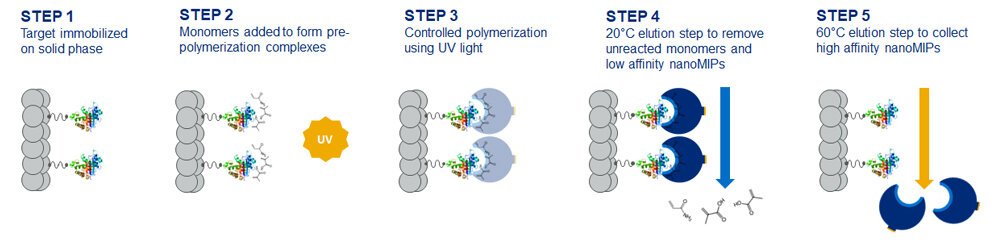
The resulting nanoMIPs are essentially a plastic cast with a 3D pocket that selectively binds the target molecule once presented to it. The binding event triggers a signal via the transducer and a signal detector displays the result to the user.
A brief history of MIPs in sensors
Whist the first mention of Molecularly Imprinted Polymers goes back almost 100 years (2), it wasn’t until the 1970’s that research into MIPs really took off. With so many potential uses, research has spanned numerous applications including purification, separation, catalysis, and drug delivery. However, it has only been in the past decade that research has really accelerated with regards to MIPs as detection agents in sensors. This is likely due to the ever-reducing size of molecularly imprinted polymers, which now commonly reach 40 – 50nm, (3) and this size is reducing further as the technology evolves. Over 30,000 papers on MIP based sensors have now been published, spanning industries including medical diagnostics, environmental monitoring, agriculture, security and food testing.
What do MIPs offer that natural receptors can’t?
STABILITY
One of the key advantages MIPs have over natural receptors is their robustness in extreme conditions. They have been proven to withstand high temperatures and pressures, as well as the presence of organic solvents. MIP Diagnostics have even sent MIPs to space and they have still performed in the same way once returned! (4) This robustness means MIPs open the door to a wider range of applications for sensors, spanning multiple industries. One such example is in the oil and gas industry, in which sampling sites are often in high temperature or high-pressure locations. Traditional biosensor receptors would perish in these types of environments, but MIP based sensors can be used on-site to improve process performance, prevent equipment corrosion, and assure high quality and purity of the end products.
GREATER VARIETY
Another advantage that MIPs offer is their ability to be generated to a wide range of targets. Traditional receptors, such as antibodies, are difficult to generate to small molecules or toxic substances but, since MIPs are completely synthetic, they do not face these challenges. Types of targets range from small molecules of less than 1nm in size, right through to whole viruses and bacteriophages of more than 400nm, demonstrated in Figure 2.
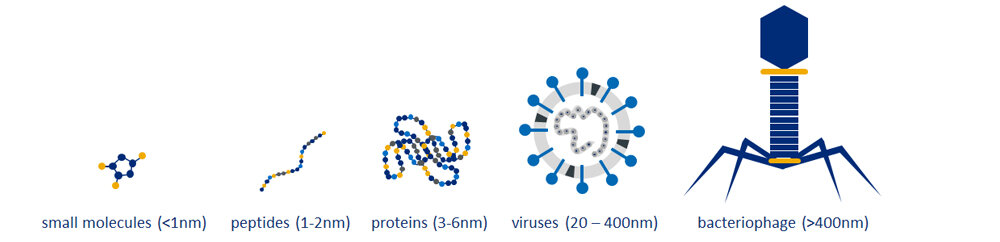
This also makes them a good choice for the development of multiplex sensors for detecting several molecules and biomarkers for improved diagnostic accuracy. R.D. Crapnell et al. have developed a MIP based multiplex sensor platform for two cardiac biomarkers, FABP and ST2, (5) and E. De Rycke et al. have developed a multiplex sensor for tracing several amphetamine synthesis markers in sewage water. (6)
EASE OF INTEGRATION
One further advantage of MIPs as the receptor in sensors is their ease of integration. Their robust characteristics means nanoMIPs enable simple and successful integration into sensor platforms when compared with natural receptors such as antibodies or nucleic acids. nanoMIPs easily couple to the solid surface of an electrode, whereas natural receptors can easily deform or denature during the coupling process.
In the context of electrochemical sensors, MIPs can be designed to produce a change of signal (current, potential, impedance) when they bind to the target molecule. As well as this, their stability and the chemistries employed means MIPs can easily be used to functionalise screen-printed electrodes for sensor development.
Alongside these features, new nanoMIPs can be developed in as little as 8 weeks. This is much more rapid than antibodies, which can take several months. Antibodies also run the risk of production issues due to underperforming cell lines, but since nanoMIPs are chemically manufactured, this issue is negated completely. This security of supply is understandably extremely attractive to commercial sensor manufacturers.
What does the future hold for MIP based sensors?
The past decade of research into MIP based sensors has laid the foundations for commercialization. As with most innovative technologies, adoption often starts in less regulated fields and molecularly imprinted polymers are no exception. Environmental monitoring and food testing are two fields in which MIP based sensors are being commercialized. Examples include a sensor to detect cigarette and marijuana smoke in hotel rooms, and a personal device to test for multiple food allergens in a meal, advising the user whether or not it is safe to consume.
Medical diagnostics is the next field in which we will see the commercialization of MIP based sensors. R&D teams are now evaluating and validating commercial molecularly imprinted polymers from companies such as MIP Diagnostics in their next wave of devices. The unique characteristics of MIPs compared with natural receptors makes them ideal for robust point of care testing and bedside analysis. We can undoubtedly expect the next decade to be the one in which MIP based sensors take their place in commercial biosensors across multiple industries.
MIP Diagnostics offers a custom development service and a growing range of off-the shelf products. Get in touch using the form below to discuss your needs with a member of our team.
References
(2) Polyakov M.V. Polyakov, 1931, Adsorption properties and structure of silica gel. Zhur Fiz Khim 2: 799–805

